David G. Mendoza-Cozatl

Associate Professor
Division of Plant Science and Technology
E-mail: mendozacozatld@missouri.edu
Office address: 271F Bond Life Sciences Center
Office phone: 573-882-1892
Plants and seeds are the main dietary source of essential nutrient metals, such as zinc (Zn), iron (Fe), manganese (Mn), and copper (Cu). However, plant-based products are also the main entry point for toxic elements, like cadmium (Cd), arsenic (As), mercury (Hg), and lead (Pb). In humans, detrimental effects of heavy metals have been linked to diabetes, hypertension, myocardial infarction, diminished lung function, and certain types of cancer.
Understanding the molecular mechanisms by which plants mobilize and accumulate heavy metals will have two major impacts on human health. First, it will enhance the nutritional value and safety of plant products by ensuring the accumulation of essential metals while avoiding the retention of toxic metals. Second, the identification of genes and molecular mechanisms that allow plants to tolerate and accumulate toxic metals will facilitate the engineering of plants for bioremediation purposes.
Our lab has three main areas of interest:
(1) Long-distance transport of heavy metals.
 (2) Plant transcriptional responses to
(2) Plant transcriptional responses to
environmental stresses.
(3) Metabolic engineering.
Long-distance Transport of Heavy Metals
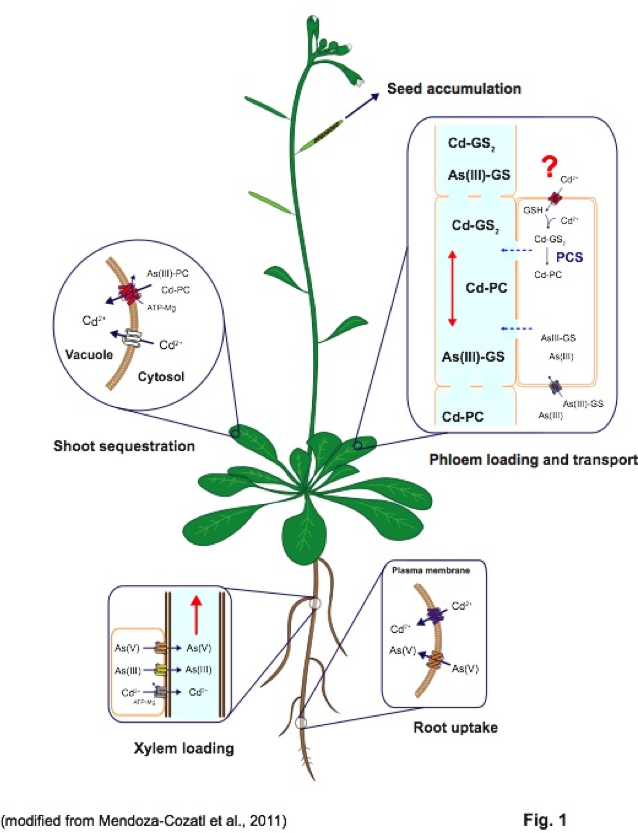
Distribution of heavy metals between roots and shoots is a dynamic process orchestrated by several plasma membrane transporters, metal-chelating molecules, xylem-loading/unloading, and phloem-loading/unloading processes. Root-to-shoot transport of metals occurs mainly through the xylem; however, due to the limited transpiration rate within reproductive tissues, the xylem plays only a minor role in allocating nutrients into the seeds. Phloem transport, on the other hand, plays a key role in delivering nutrients, including metals, to developing seeds. Our lab investigates the isolation, identification, and characterization of phloem-specific transporters of trace metals (essential and toxic) using the reference plant Arabidopsis thaliana.
Plant Transcriptional Responses to Environmental Stresses
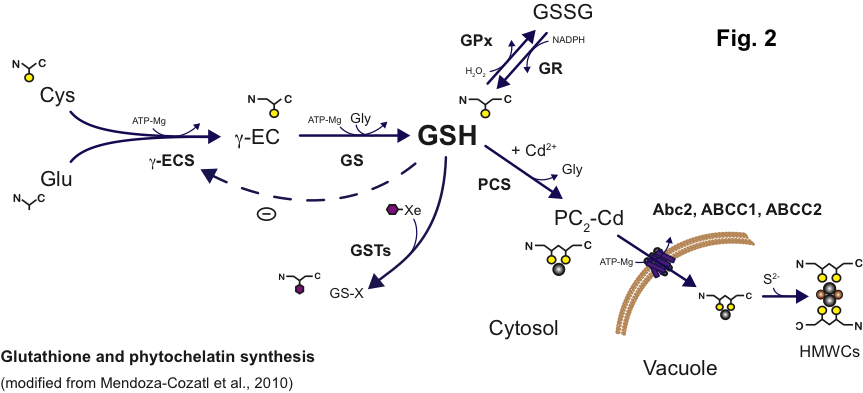
Glutathione (GSH) and phytochelatin biosynthesis is perhaps the most studied mechanism for heavy metal detoxification and accumulation in plants. GSH has several functions in the cell, including the control of cellular redox, detoxification of xenobiotics, sulfur storage, synthesis of glucosinolates, and synthesis of metal-chelating peptides called phytochelatins (PCs). The enzymes that synthesize GSH have been identified, and their kinetic properties well established. The transcriptional regulation of GSH biosynthetic genes, however, is largely unknown. Using a systems biology approach, we are working to identify transcriptional regulators of GSH biosynthesis in an effort to understand how plants sense and respond to different environmental stresses.
Metabolic Engineering
Our lab is also exploring ways to increase the capacity of plants to accumulate toxic compounds for bioremediation purposes. Metabolic engineering requires a deep understanding of the genetic and regulatory processes that govern the structure of metabolic pathways. Traditionally, regulation of metabolic pathways is attributed to rate-limiting steps. However, the “rate-limiting” concept can be misleading since it assumes that only one enzyme controls both, the rate of synthesis of a given metabolite as well as its concentration. Our goal is to move away from the rate-limiting step concept and understand metabolic pathways as a whole. Using kinetic modeling, overexpression and tissue-specific expression of enzymes and transporters we aim to manipulate the capacity of plants to tolerate and accumulate compounds in specific tissues such as roots, shoots or seeds.
- National Science Foundation CAREER Award (2012)
- 2010 Cozzarelli Prize, Applied Biological, Agricultural, and Environmental Sciences
(National Academy of Sciences, USA).
- 2006-2008 PEW Latin American Fellowship in Biomedical Sciences, PEW Foundation.
- 2005 Weizmann award for the best Ph.D. thesis, nationwide, in Natural Sciences
(Mexican Academy of Sciences).
Khan MA, Castro-Guerrero NA, McInturf SA, Nguyen NT, Dame AN, Wang J, Bindbeutel RK, Joshi T, Jurisson SS, Nusinow DA, Mendoza-Cózatl DG. (2018) Changes in iron availability in Arabidopsis are rapidly sensed in the leaf vasculature and impaired sensing leads to opposite transcriptional programs in leaves and roots. Plant Cell Environ. 41:2263-76.
Acosta-Gamboa LM, Liu S, Langley E, Campbell Z, Castro-Guerrero4 NA, Mendoza-Cózatl DG, Lorence A. (2017) Moderate to severe water limitation differentially affects the phenome and ionome of Arabidopsis. Funct. Plant Biol. 44, 94–106.
Zhang Z, Xie Q, Jobe TO, Kau AR, Wang C, Li Y, Qiu B, Wang Q, Mendoza-Cózatl DG, Schroeder JI (2016). Identification of AtOPT4 as a Plant Glutathione Transporter. Mol Plant. 9:481-4.
Mendoza-Cózatl DG, Xie Q, Akmakjian GZ, Jobe TO, Patel A, Stacey MG, Song L, Demoin DW, Jurisson SS, Stacey G, Schroeder JI. (2014) OPT3 is a component of the iron-signaling network between leaves and roots and misregulation of OPT3 leads to an over-accumulation of cadmium in seeds. Mol Plant. 7:1455-69.
Mendoza-Cózatl DG, Jobe TO, Hauser F, Schroeder JI. Long-distance transport, vacuolar sequestration, tolerance, and transcriptional responses induced by cadmium and arsenic. Current Opinion in Plant Biology. Article in Press.
Mendoza-Cózatl DG, Zhai Z, Jobe TO, Akmakjian GZ, Song WY, Limbo O, Russell MR, Kozlovskyy VI, Martinoia E, Vatamaniuk OK, Russell P, Schroeder JI. Tonoplast-localized Abc2 transporter mediates phytochelatin accumulation in vacuoles and confers cadmium tolerance. Journal of Biological Chemistry 2010;285(52):40416-40426.
Song WY, Park J, Mendoza-Cózatl DG, Suter-Grotemeyer M, Shima D, Hörtensteiner S, Geisler M, Weder B, Rea PA, Rentsch D, Schroeder JI, Lee Y, Martinoia E. Arsenic tolerance in Arabidopsis is mediated by two ABCC-type phytochelatin transporters. Proceedings of the National Academy of Sciences of the United States of America 2010;107(49):21187-21192.
Sung DY, Kim TH, Komives EA, Mendoza-Cózatl DG, Schroeder JI. ARS5 is a component of the 26S proteasome complex, and negatively regulates thiol biosynthesis and arsenic tolerance in Arabidopsis. Plant Journal 2009;59(5):802-812.
García-García JD, Rodríguez-Zavala JS, Jasso-Chávez R, Mendoza-Cozatl D, Moreno-Sánchez R. Chromium uptake, retention and reduction in photosynthetic Euglena gracilis. Archives of Microbiology 2009;191(5):431-440.
Estrella-Gómez N, Mendoza-Cózatl D, Moreno-Sánchez R, González-Mendoza D, Zapata-Pérez O, Martínez-Hernández A, Santamaría JM. The Pb-hyperaccumulator aquatic fern Salvinia minima Baker, responds to Pb2+ by increasing phytochelatins via changes in SmPCS expression and in phytochelatin synthase activity. Aquatic Toxicology 2009;91(4):320-328.
Mendoza-Cózatl DG, Butko E, Springer F, Torpey JW, Komives EA, Kehr J, Schroeder JI. Identification of high levels of phytochelatins, glutathione and cadmium in the phloem sap of Brassica napus. A role for thiol-peptides in the long-distance transport of cadmium and the effect of cadmium on iron translocation. Plant Journal 2008;54(2):249-259.
Alvarez-Legorreta T, Mendoza-Cozatl D, Moreno-Sanchez R, Gold-Bouchot G. Thiol peptides induction in the seagrass Thalassia testudinum (Banks ex König) in response to cadmium exposure. Aquatic Toxicology 2008;86(1):12-19.
García-Ríos V, Freile-Pelegrín Y, Robledo D, Mendoza-Cózatl D, Moreno-Sánchez R, Gold-Bouchot G. Cell wall composition affects Cd2+ accumulation and intracellular thiol peptides in marine red algae. Aquatic Toxicology 2007;81(1):65-72.
Mendoza-Cózatl DG, Rodríguez-Zavala JS, Rodríguez-Enríquez S, Mendoza-Hernandez G, Briones-Gallardo R, Moreno-Sánchez R. Phytochelatin-cadmium-sulfide high-molecular-mass complexes of Euglena gracilis. FEBS Journal 2006;273(24):5703-5713.
Mendoza-Cózatl DG, Rangel-González E, Moreno-Sánchez R. Simultaneous Cd2+, Zn2+, and Pb2+ uptake and accumulation by photosynthetic Euglena gracilis. Archives of Environmental Contamination and Toxicology 2006;51(4):521-528.
Mendoza-Cózatl DG, Moreno-Sánchez R. Control of glutathione and phytochelatin synthesis under cadmium stress. Pathway modeling for plants. Journal of Theoretical Biology 2006;238(4):919-936.
Avilés C, Torres-Márquez ME, Mendoza-Cózatl D, Moreno-Sánchez R. Time-course development of the Cd2+ hyper-accumulating phenotype in Euglena gracilis. Archives of Microbiology 2005;184(2):83-92.
Jasso-Chávez R, García-Cano I, Marín-Hernández A, Mendoza-Cózatl D, Rendón JL, Moreno-Sánchez R. The bacterial-like lactate shuttle components from heterotrophic Euglena gracilis. Biochimica et Biophysica Acta - Bioenergetics 2005;1709(2):181-190.
Mendoza-Cózatl D, Loza-Tavera H, Hernández-Navarro A, Moreno-Sánchez R. Sulfur assimilation and glutathione metabolism under cadmium stress in yeast, protists and plants. FEMS Microbiology Reviews 2005;29(4):653-671.
Mendoza-Cózatl DG, Moreno-Sánchez R. Cd2+ transport and storage in the chloroplast of Euglena gracilis. Biochimica et Biophysica Acta - Bioenergetics 2005;1706(1-2):88-97.
Mendoza-Cozatl D, Devars S, Loza-Tavera H, Moreno-Sánchez R. Cadmium accumulation in the chloroplast of Euglena gracilis. Physiologia Plantarum 2002;115(2):276-283.